Problem 47 Read Isotope File Isotopes Chemical Element Ground State Number Protons Differ Q43803448

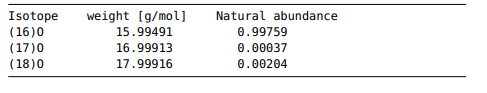

Problem 4.7. Read isotope file Isotopes of a chemical element in its ground state have the same number of protons but differ in the number of neutrons. The weight of isotopes of the same chemical element will therefore be different. The molar mass, M, of a chemical element, can be calculated by summing over all its isotopes M = mwi, where mi is the weight of the i-th isotope and w; the corresponding natural abundance. The file Oxygen.txt, which is given below, contains the information on Oxygen’s isotopes (180, ’70 and 180). Isotope (16)0 (17)0 (18)0 weight (g/mol) 15.99491 16.99913 17.99916 Natural abundance 0.99759 0.00037 0.00204 Write a script in Python to read the file Oxygen.txt and extract the weights and the natural abundance of all the isotopes of Oxygen. Use these to calculate the molar mass of Oxygen. Print out the result with four decimals and provide the correct units. Show transcribed image text Problem 4.7. Read isotope file Isotopes of a chemical element in its ground state have the same number of protons but differ in the number of neutrons. The weight of isotopes of the same chemical element will therefore be different. The molar mass, M, of a chemical element, can be calculated by summing over all its isotopes M = mwi, where mi is the weight of the i-th isotope and w; the corresponding natural abundance. The file Oxygen.txt, which is given below, contains the information on Oxygen’s isotopes (180, ’70 and 180).
Isotope (16)0 (17)0 (18)0 weight (g/mol) 15.99491 16.99913 17.99916 Natural abundance 0.99759 0.00037 0.00204
Write a script in Python to read the file Oxygen.txt and extract the weights and the natural abundance of all the isotopes of Oxygen. Use these to calculate the molar mass of Oxygen. Print out the result with four decimals and provide the correct units.
Expert Answer
Answer to Problem 4.7. Read isotope file Isotopes of a chemical element in its ground state have the same number of protons but di…
OR